
Dự kiến trong tuần sau, Việt Nam sẽ bắt đầu thử nghiệm giai đoạn 3 vắc xin Nanocovax với khoảng 13.000 tình nguyện viên.
Hội đồng Đạo đức của Bộ Y tế vừa họp, xem xét thông qua quyết định cho phép triển khai nghiên cứu lâm sàng giai đoạn 3 vắc xin ngừa Covid-19 Nanocovax.
Nguồn tin từ Bộ Y tế cho biết, về cơ bản hội đồng đã đồng ý nghiên cứu giai đoạn 3 đa trung tâm, tuy nhiên đơn vị nghiên cứu cần hoàn chỉnh hồ sơ trong tuần này, trình Bộ Y tế để ký duyệt trước khi triển khai.
Trong giai đoạn 3, vắc xin Nanocovax sẽ thử nghiệm đa trung tâm tại các cơ sở trong nước với khoảng 13.000 tình nguyện viên. Tại miền Bắc có Học viện Quân y và Hưng Yên , khu vực phía Nam có Viện Pasteur TP. HCM và Long An tham gia.
Giai đoạn 3 sẽ chia thành nhiều giai đoạn nhỏ, trước mắt sẽ tiêm cho 800 tình nguyện viên đã đăng ký, sau đó sẽ tiếp tục tuyển thêm.
Liều tối ưu được lựa chọn cho giai đoạn 3 là 25mcg. Qua nghiên cứu, cả 3 nhóm liều 25mcg, 50mcg và 75mcg đều sinh miễn dịch tương đương, do hiệu quả ngang nhau nên Hội đồng Đạo đức chọn liều thấp nhất để sản xuất, tiết kiệm nguyên liệu.
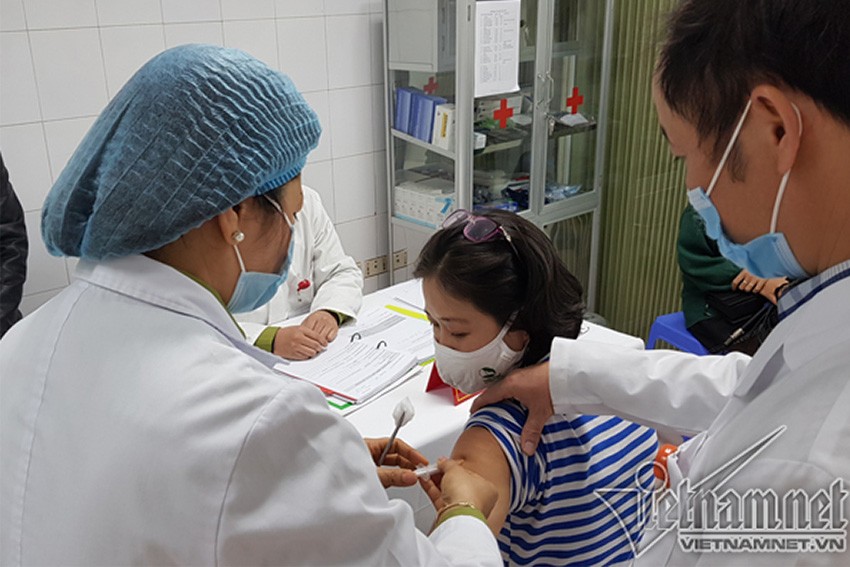 Chị N. là một trong 3 tình nguyện viên đầu tiên tham gia thử nghiệm vắc xin Nanocovax. Ảnh: Thúy Hạnh
Chị N. là một trong 3 tình nguyện viên đầu tiên tham gia thử nghiệm vắc xin Nanocovax. Ảnh: Thúy Hạnh
Thử nghiệm giai đoạn 2 vắc xin Nancocovax đã tiêm xong từ ngày 8/4, tuy nhiên việc theo dõi sẽ kéo dài trong 1 năm. Hiện đang là giai đoạn đánh giá giữa kỳ nhưng kết quả nghiên cứu rất khả quan, vắc xin sinh miễn dịch rất tốt và có hiệu quả với cả biến thể Anh và Nam Phi. Tác dụng với biến thể Ấn Độ sẽ tiếp tục được nghiên cứu trong giai đoạn 3.
Nếu thuận lợi, đến cuối tháng 9, thử nghiệm giai đoạn 3 sẽ hoàn tất, khi đó Việt Nam có thể sản xuất vắc xin ngừa Covid-19 đầu tiên.
Theo các nghiên cứu trên thế giới, mức độ đáp ứng miễn dịch của tình nguyện viên được đánh giá tốt khi sản sinh ra kháng thể gấp 4 lần so với bình thường, cá biệt có trường hợp gấp 20 lần.
Tuy nhiên thực tế với Nanocovax, sau tiêm mũi đầu tiên 35 ngày, lượng kháng thể trên một số tình nguyện viên tăng lên đến hơn 60 lần, sau 3 tháng vẫn còn 34 lần. Đây là con số rất ấn tượng.
Vắc xin Nanocovax do công ty Nanogen sản xuất dựa trên công nghệ tái tổ hợp protein. Thử nghiệm lâm sàng giai đoạn 1 bắt đầu từ ngày 17/12/2020, đến ngày 8/4 vừa qua đã kết thúc giai đoạn 2 trên 554 tình nguyện viên, trong đó có 108 người cao tuổi.
Trong giai đoạn 2, các tình nguyện viên được chia 4 nhóm, tiêm 3 mức liều 25mcg, 50mcg, 75mcg và nhóm còn lại tiêm giả dược.
Kết quả giai đoạn 2 cho thấy, cả 3 mức liều đều đảm bảo an toàn, 100% người tiêm đều sinh miễn dịch ở các mức độ khác nhau.
Trong đó, chỉ số kháng thể trung hòa virus (khả năng tiêu diệt virus SARS-CoV-2) sau 2 tuần tiêm mũi 2 ở nhóm liều 25mcg cho kết quả cao nhất, với trên 90%, 2 nhóm liều 50mcg và 75mcg cho kết quả thấp hơn không đáng kể.
Nanocovax là vắc xin ngừa Covid-19 đầu tiên của Việt Nam thử nghiệm lâm sàng. Vắc xin thứ 2 trong nước tham gia "cuộc đua" là Covivac của công ty IVAC đã tiêm xong giai đoạn 1.
Dự án Nanocovax có thể nhận được tài trợ hàng trăm triệu đô la
Theo nguồn tin đáng tin cậy, dự án Nanocovax nhận được tài trợ lên tới hàng trăm triệu đô la Mỹ từ một tập đoàn tư nhân trong nước. Khoản tài trợ này nhằm thúc đẩy quá trình thử nghiệm vắc xin nội giai đoạn cuối cùng để phê duyệt sử dụng cho toàn dân.
Đồng thời, hệ thống sản xuất, vật tư thiết bị đang khẩn trương đầu tư nhằm cung ứng đủ số lượng vắc xin, bổ sung với số lượng vắc xin nhập khẩu. Mục đích lớn của chương trình tài trợ này là hoàn thành chương trình tiêm chủng vắc xin phòng chống Covid-19 cho người dân Việt Nam ngay trong năm 2021.
Nhà tài trợ và đơn vị nghiên cứu, sản xuất vắc xin nội hy vọng dự án Nanocovax sẽ góp phần cho Việt Nam mở cửa kinh tế, tiếp tục là một điểm sáng về phòng chống dịch Covid-19 trên bản đồ thế giới, một điểm đến an toàn cho đầu tư, du lịch, giao thương và mọi nhu cầu đi lại, giao lưu của mọi người dân và du khách.
Nguồn: https://vietnamnet.vn/
Tin liên quan
- Triển lãm Y tế Việt Nam lần thứ 12 chính thức khởi động
- Nhật thử nghiệm thuốc Covid-19 ngày uống một viên
- Trên 600 gian hàng quy tụ tại Triển lãm Y tế quốc tế Việt Nam 2017
- Bệnh viện K ứng dụng nội soi nhuộm màu phóng đại trong chẩn đoán, điều trị ung thư thực quản sớm
- Vietnam’s Growing Pharmaceutical Industry
